|
11/12/2023 0 Comments Periodic table s element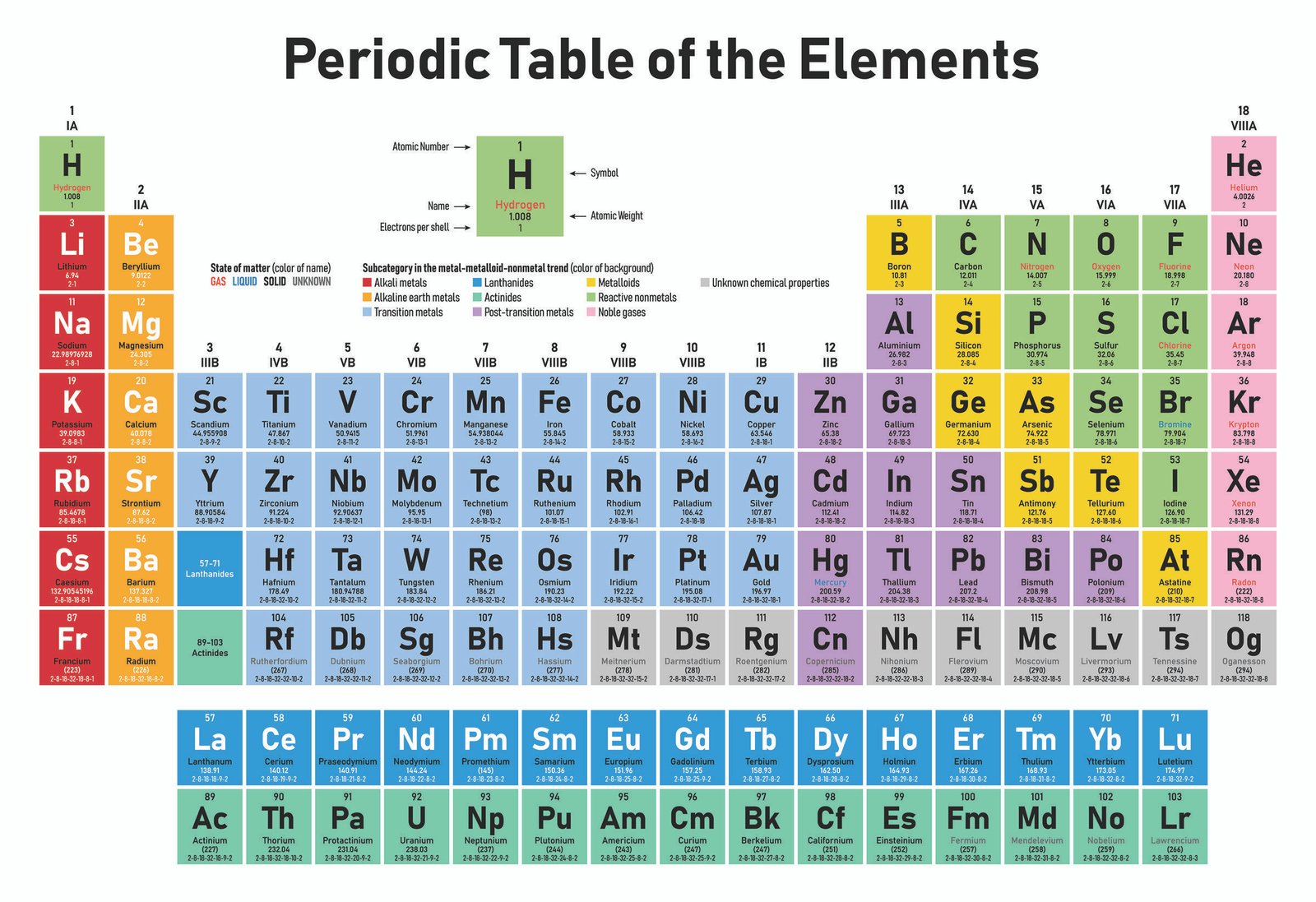
Also, the increasing order of the atomic mass of the elements wasn’t regular throughout the table. Some of them are, that it failed to designate Hydrogen and Noble gases in it. However, Mendeleev’s Periodic Table fails due to many flaws in it. In the Mendeleev periodic table, the horizontal row and vertical columns were referred to as groups and periods, respectively. Only 63 elements were known at the time of Mendeleev’s work. In order to represent similarities and trends in the behavior of elements, Mendeleev developed the periodic table, which is an arrangement of elements in an increasing atomic mass order in tabular form.Īccording to Mendeleev’s Periodic Table, elements were arranged as per their fundamental property, atomic mass, and chemical characteristics. The Mendeleev periodic table was the most important of many periodic tables like the Newlands Octave Law that was rejected in 1869. Russian scientist Dmitri Ivanovich Mendeléev made the most significant contribution to the early creation of the periodic table. The elements listed are arranged according to the increasing order of atomic number and their respective atomic weight, symbols, density, and electronegativity. Here is the table representing 118 elements of the periodic table. Such elements are present in Lanthanides and Actinides groups. f-block elements: These elements have the last electron filled in their f-subshell.d-block elements are also called transition elements as they have partially filled d-orbitals in their ground state. 
These elements have the last electron filled in their d-subshell. d-block elements : The elements present in groups 3 to 12 are d-block elements.The elements included in this block from different groups are termed as Boron Family (Group 13), Carbon Family (Group 14), Nitrogen Family (Group 15), Oxygen Family (Group 16), and Fluorine Family (Group 17). These elements have the last electron filled in their p-subshell. p-block elements : The elements included in groups 13 to 17 are p-block elements.The elements included in the s-block are Alkali metals and Alkaline Earth Metals. s-block elements: The first and the second group elements that have the last electron filled in the s-subshell are called s-block elements.Further, the elements are divided into different blocks. The 118 elements are arranged in 7 periods and 18 groups as shown below. Classification of Elements in the Periodic Table For example properties of particular elements such as their mass, electron number, electron configuration, and their unique chemical properties. The periodic table is an essential aspect of Chemistry because it is an arrangement of all the known elements and therefore provides information about elements and their relation with one another in one easy-to-use reference. Role of Mahatma Gandhi in Freedom Struggle.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
